CH391L/S14/BioBricks
Contents |
What are BioBricks, and what are their purpose?
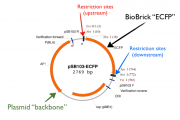
A BioBrick is a sequence of DNA with a predefined structure and function. The information is held in a plasmid, or a circular piece of DNA that can be inserted, and replicated into bacteria.
Before BioBricks, there was little standardization in assembly techniques for DNA sequences. It was necessary to create a standardized system of biological building blocks to enable scientists to go beyond the experiment of DNA assembly and allow them to focus on more complicated research. BioBricks were created as a set of standard and interchangeable “parts” that could be assembled into sub-components. [1] According to the Registry of Standard Biological Parts, which contains DNA for thousands of previously created parts submitted by iGEM members, a standard biological part is “a functional unit of DNA that encodes for a specific biological function. Parts have been standardized so they can be used to efficiently develop biological systems in living cells." [2]The advantage of BioBricks is that the assembly of these parts could be outsourced to others and new experimentation could rely heavily on previously manufactured components. The BioBricks foundation was co-founded in 2005 by Drew Endy. Tom Knight and Drew Endy also helped co-found the Registry of Standard Biological Parts.
The creation of a standard sequence interface for all BioBricks:
The original BioBrick assembly standard created by Tom Knight:
In order to easily join and manipulate segments of DNA, BioBrick assembly standards requires the use of defined prefix and suffix sequences that flank both sides of the BioBrick. These prefix and suffix regions (such as EcoRI, NotI, XbaI, SpeI, and PstI) can be cut by specific restriction endonucleases. Restriction endonucleases are a key element in the cutting and conjoining of segments of DNA, and will be elaborated in further detail in a later section.
The upstream end (Prefix) contains vector insert restriction sites with the following sequence:
5' --gca GAATTC GCGGCCGC T TCTAGA G --- 3'
3' --cgt CTTAAG CGCCGGCG A ACATCT C --- 5'
EcoRI NotI XbaI
The downstream end (Suffix) contains vector insert restriction sites with the following sequence:
5' --- T ACTAGT A GCGGCCG CTGCAG gct--- 3'
3' --- A TGATCA T CGCCGGC GACGTC cga--- 5'
SpeI NotI PstI
When combined, the entire component vector looks like this:
5’ --gca GAATTC GCGGCCGC T TCTAGA --insert--T ACTAGT A GCGGCCG CTGCAG gct--
3' --cgt CTTAAG CGCCGGCG A ACATCT -------- A TGATCA T CGCCGGC GACGTC gca--
EcoRI NotI XbaI SpeI NotI PstI
The component vector (the insert) cannot contain any of the restriction sites to avoid any unwanted cutting by restriction enzymes, and any such sites are removed by point mutations. [1]
Restriction Endonucleases
Restriction enzymes, or restriction endonucleases are enzymes that cleave DNA at restriction sites. In bacteria, restriction enzymes are used as a defense mechanism against invading viruses. A restriction site is a sequence of base pairs (generally 4-8 base pairs in length) that is recognized and spliced by a restriction enzyme. To cut DNA, restriction enzymes make two incisions: once through each side of the sugar-phosphate backbone. [3] Restriction enzymes can cut in two ways: there are blunt end cutters or sticky end cutters. Blunt ends are where the enzymes cut both strand of the target DNA at the same spot creating blunt ends. Sticky end cutters cut both strand of the target DNA at different spots creating 3'- or 5'-overhangs of 1 to 4 nucleotides (so-called sticky ends). [4] There are 4 kinds of restriction enzymes.
Types of Restriction Endonucleases
Type I:
These were the first type of enzymes to be discovered, but have very little use in synthetic biology because they randomly cleave the DNA varying distances from the restriction site. The recognition site is composed of two different, asymmetrical portions: one containing 3-4 nucleotides, and another containing 4-5 nucleotides, and are separated by a non-specific spacer that is composed of 6-8 nucleotides. They are a multifunctional protein with both methylase and restriction activity. [3] [5]
Type II:
These are the most commonly available and the restriction enzymes used in DNA analysis. They are a homodimer, meaning that they are composed of two identical molecules. They do not use ATP for their activity, but rather they generally utilize magnesium. Type II restriction enzymes recognize palindromic sequences, which are sequences that are read the same 5’ as they are 3’. They produce discrete restriction fragments and gel banding patterns, which is why they are the most useful in the laboratory. New enzymes from this family have been discovered, but do not follow all classical criteria of type II enzymes. Subgroups have been created to categorize the enzymes that deviate from the criteria. Type II enzymes are a single function restriction enzyme and solely cleave but do not methylate. [3][6]
Type III:
This type of restriction enzyme recognize two separate non-palindromic sequences that are inversely oriented. They cleave DNA 20-30 base pairs from the restriction site. They are composed of more than one subunit and require ATP, but do not hydrolyze it. They require the presence of unmethylated regions that are inversely oriented for restriction to occur. They are found in prokaryotic organisms, and are a useful component in the defense against foreign invading DNA. [7]
Type IV
Type IV enzymes recognize modified, typically methylated DNA and are exemplified by the McrBC and Mrr systems of E. coli.
The Role of Ligase
Ligase is an enzyme that facilitates and catalyzes the creation of phosphodiester bonds between two pieces of DNA that have been cleaved by restriction enzymes. In bacteria, ligase is used to connect the two ends of a newly synthesized circular piece of DNA. In synthetic biology, ligase is utilized to connect two pieces of DNA that did not previously exist together, which is the case in fixing a BioBrick into a plasmid vector.
Considerations in the selection of Restriction Enzymes
According to Tom Knight,
“The choice of restriction enzymes was a significant issue in the design of the pSB103 vector and assembly plan. We wanted restriction enzymes which were easy to use and reliable, which functioned in compatible buffer systems and at compatible temperatures, which could be heat killed, provided complete digestion, with few required bases outside of their recognition site, and exhibited low star activity. In addition, we wanted four base overhangs to enhance ligation efficiency.”
Another issue that was encountered was the sequence recognition site. There were several factors that had to be considered such as avoiding the accidental creation of ATG start codons in strange places, and also avoiding the creation of methylation sensitive sites. [1]
List of Restriction Enzymes used with BioBricks
| Enzyme | Source | Recognition Sequence | Cut |
|---|---|---|---|
| EcoRI | Escherichia coli |
5'GAATTC 3'CTTAAG |
5'---G AATTC---3' 3'---CTTAA G---5' |
| NotI | Nocardia otitidis |
5'GCGGCCGC 3'CGCCGGCG |
5'---GC GGCCGC---3' 3'---CGCCGG CG---5' |
| SpeI | Sphaerotilus natans |
5'ACTAGT 3'TGATCA |
5'---A CTAGT---3' 3'---TGATC A---5' |
| XbaI | Xanthomonas badrii |
5'TCTAGA 3'AGATCT |
5'---T CTAGA---3' 3'---AGATC T---5' |
| PstI | Providencia stuartii |
5'CTGCAG 3'GACGTC |
5'---CTGCA G---3' 3'---G ACGTC---5' |
BioBrick composition techniques
There are two methods of composing a biobrick component: Prefixing one component to another, or postfixing one component with another. Both techniques result in a new, compound component, which can then be used as either an insert or vector in other reactions.
Prefixing a recipient vector with a donor component
In order to prefix a recipient component, a front vector is created. To do so, the component is cut with EcoRI and XbaI enzymes:
5' --gca G *CTAGA G---- 3'
3' --cgt CTTAA* T C---- 5'
EcoRI XbaI
EcoRI and SpeI are used to cut the donor component, creating a front insert:
5' *AATTC GCGGCCGC T TCTAGA G --Insert-- T A 3'
3' G CGCCGGCG A ACATCT C --Insert-- A TGATC* 5'
EcoRI NotI XbaI SpeI
The EcoRI sites and mixed SpeI/XbaI sites are ligated creating this segment:
5' --gca G *AATTC GCGGCCGC T TCTAGA G--insert--T A *CTAGA G---- 3'
3' --cgt CTTAA* G CGCCGGCG A ACATCT C--insert--A TGATC* T C---- 5'
EcoRI NotI XbaI Mixed
The EcoRI and XbaI sites are recreated, and a mixed, uncuttable SpeI/XbaI site is created. The other restriction regions SpeI and PstI remain untouched. [1]
Postfixing a recipient vector with a donor component
A back vector is created with SpeI and PstI to insert a donor fragment after a component on a vector. This vector is created:
5' --T A *G gct--- 3'
3' --A TGATC* ACGTC cga--- 5'
SpeI PstI
The donor component is cut with XbaI and PstI to create a back insert:
5' *CTAGA G --insert-- T ACTAGT A GCGGCCG CTGCA 3'
3' T C --insert-- A TGATCA T CGCCGGC G* 5'
XbaI SpeI NotI PstI
Similar to the Prefixing method, the two products are ligated together. The SpeI and PstI sites are restored, while there is an uncuttable, mixed SpeI/XbaI site that is made at the junction of the two inserts. In contrast to the Prefixing method, now the EcoRI and XbaI sites remain untouched in the upstream region: [1]
5' --T A *CTAGA G --insert-- T ACTAGT A GCGGCCG CTGCA *G gct--- 3'
3' --A TGATC* T C --insert-- A TGATCA T CGCCGGC G* ACGTC cga--- 5'
Mixed SpeI NotI PstI
Method of assembling multiple BioBrick parts
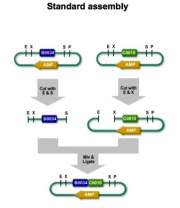
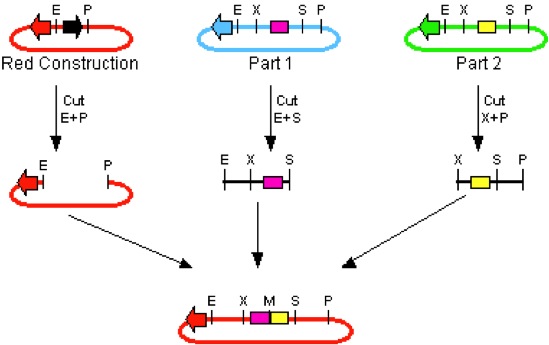
In standard assembly, more than one BioBrick can be added to a single plasmid vector. This can be achieved by methodic utilization of the restriction enzymes.
Following along with the example photo on the left, where E=EcoRI, X=XbaI, S=SpeI, and P=PstI, it is shown that in order to insert the two parts, the region between E and P must be cut on the plasmid vector. To insert two different DNA strands, they must be cut so that the restriction sites can line up and be ligated together. As a result Part I is cut so that the E regions match up, and the S and X regions connect to make a mixed product. In Part II, the DNA is cut so that the P regions match up, and again cut so that the S and X regions between Part I and II can make a mixed product.
Laboratory methods
To combine two different BioBricks together in a plasmid:
- 1) Three PCR tubes are needed, one for the destination plasmid, one for the upstream part, and one for the downstream part.
- 2) To each tube, water and the individual part is added. NEBuffer is then added, along with the appropriate restriction enzyme for each part.
- 3) The parts are digested.
- 4) The parts are then mixed together with DNA ligase buffer and incubated.
If done correctly, transformation will have occurred. A more detailed description of assembly can be found at GinkgoBioWorks
Future Directions
While BioBricks are being utilized by iGEM teams and synthetic biologists, some fundamental issues have been identified with the BioBricks system, namely, none of the restriction sites can be present in any part of the BioBrick DNA. This limits synthetic biologists in what they can create, especially when trying to make longer DNA sequences since the incidence of accidentally having a restriction site in said DNA is more likely. Many iGEM teams, (including the UT Austin iGEM team) are proposing new methods to fix the component DNA within the plasmid vector. The UT iGEM team proposed a new method, the general idea being this: to create an expanded recognition sequences to reduce the likelihood of the recognition sequence occurring within the BioBrick. This eliminates the need for site directed mutation that could potentially alter the function of the BioBrick.
References
- T. Knight, “Idempotent Vector Design for Standard Assembly of Biobricks” (MIT, Cambridge, MA, 2003).
Information regarding Biobrick, origins, standards, and composition techniques - http://parts.igem.org/Help:Parts
Information on the basics of BioBricks - Roberts R, and Murray K (1976) Restriction Endonuclease "Critical Reviews in Biochemistry and Molecular Biology" 4:123-164
Restriction endonuclease function of class I and II enzymes - Sambrook, J., Fritsch, E.F. & Maniatis, T. (1989) Molecular Cloning: A laboratory manual Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY 2:1.63-1.70
Sticky and blunt ends of restriction enzymes - Murray N (2000) Type I Restriction Systems: Sophisticated Molecular Machines "Microbiol. Mol. Biol. Rev." 2000 64:412-434
Type I restriction endonuclease details - Jeltsch A, Pingoud A (2001) Structure and function of type II restriction endonucleases "Nucleic Acids Research" 29(18):3705–3727
In-depth information on structure and function of Type II restriction enzyme - Rao D, Dryden D, Bheemanaik S (2013) Type III restriction-modification enzymes: a historical perspective "Nucleic Acids Research" 2014;42:56-69
Type III restriction endonuclease functional information