Microbe Hackers
Contents |
Welcome to the Microbe Hackers!
IMPORTANT NOTE: We are beginning a complete update for this site in September 2022. This line will be updated once it starts, and this line will be removed once the update is finished. delete this
IMPORTANT NOTE: We are currently updating this site. This line will be removed once the update is finished.
Tarun has started editing this page 9/30
Note: This website has been optimized for laptop displays, and may look a bit odd on mobile devices or larger monitors.
The Microbe Hackers is a very active and exciting undergraduate teaching and research lab at the University of Texas at Austin. We are part of the Freshman Research Initiative (the FRI program). We conduct research in the field of synthetic biology during the academic year as well as the summer. The stream is lead by Professor Dennis Mishler, the research educator, and Professor Barrick, the stream's PI. On our page below, you will find information about our research projects, our course curriculum, and what it means to be a "Microbe Hacker" and a member of the FRI program overall.
In this FRI stream, we do research in the vast and emerging field of synthetic biology. We genetically engineer bacteria for a purpose of our choosing. Information about our projects can be found below. Briefly, in order to modify or engineer our bacteria for some of the applications below, we must work with "genetic devices" (DNA sequences) and specific technology and techniques that are used in research across many disciplines: synthetic biology, molecular biology, microbiology, cell biology, and many more.
Students in our group have presented their research at symposiums on campus, conferences across the country, published blog posts and research articles, and have gone on to graduate programs in the health professions and business school, medical school, and PhD programs. Others have taken their experience and pivoted, using the time with us to develop vital skills for 21st century job markets. Many of these students, maybe even most, joined us with little to NO previous lab experience.
Are you a future Microbe Hacker?
If you are interested in research, are looking for a challenge, and you think synthetic biology is something interesting, then YES!
You sound like the perfect Microbe Hacker! Being a Microbe Hacker is about doing research, but more importantly, about developing your potential and preparing you for whatever awaits you in the future.
For more information regarding joining the Microbe Hackers, please read below in the "open house" section.
Stream Research
In the Microbe Hackers lab, our research projects are conducted with input from our students. Our Cyanobacteria project was initiated by advanced undergraduates in 2016 and has continued as a project that our students work on since then. Other projects were initially conceived by Professor Mishler or Professor Barrick, but have been worked on extensively (and sometimes exclusively!) by undergraduates in the group. Currently, we have four active areas of research. Each of these projects is described below, highlighting recent progress, student accomplishments, and our next steps.
Caffeinated Coli
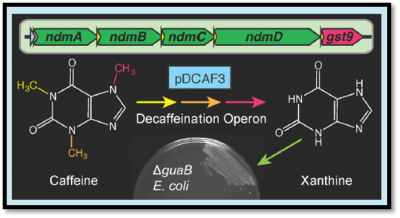
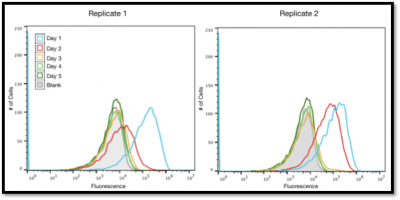
The Caffeinated Coli project has been part of the Microbe Hackers group since before we existed. These bacteria have been modified to be "addicted" to caffeine. If they don't have caffeine, they can't grow! However, if you give them caffeine, they become active and grow until they metabolize all of the caffeine present. After growing with caffeine, we can count the final number of cells grown to determine how much caffeine was originally present. This "bioassay" is very accurate, and we have used it to calculate the amount of caffeine in a cup of coffee, a can of coke, or an energy drink.
The original, "Version 1.0" was pioneered by the UT Austin iGEM team with Professor Barrick. This story was published in 2013. Since then, the Microbe Hackers have continued to work on Caffeinated Coli. Our "Version 2.0" has improved upon the original assay by creating a suite of plasmids that can specifically convert various methylxanthines: caffeine, theophylline, theobromine, etc... into xanthine. Thus, the bioassay can now determine not just how much caffeine is in your drink, but also how much of each of the other methylxanthines are also present. In fact, the bioassay is as good as standard chemical methods, such as HPLC. The "Version 2.0" story was submitted for publication at the end of August 2019 and was accepted for publication in September 2019. This paper contains five undergraduate authors: Three alumni now in either a PhD program, Medical, or Pharmacy school and two current Microbe Hackers.
We are now working on "Version 3.0" of the bioassay, attempting to improve assay sensitivity and growth conditions, while demonstrating its usage on a wider array of samples, including environmental and biological samples. These experiments are being conducted by current Microbe Hackers. Two of our current students, Mina K. and David G., will present their research at the annual Fall Undergraduate Research Symposium here at UT Austin on September 28th. Previous research can be found covering the creation of caffeinated coli, its use by our students in Austin, and an initial Version 2.0 attempt as part of iGEM 2015 in complex beverages.
Caffeine Kit
Note: This section was just created by Tarun and needs to be checked for accuracy.
This project was started during COVID-19 as a virtual project. The goal then was to figure out the logistics of creating an educational kit that high schoolers could use to learn synthetic biology using the pDCAF strains created in the Caffeinated Coli project. Since COVID-19, the project is currently finalizing the materials that will be sent with the kit, such as materials list, procedures, etc. The researchers are also ensuring that the experiments work as expected and that the materials sent in the kit are good quality.
Next steps include finishing the educational material so it is more thorough and easy for hgih schoolers to understand. Since spectrophotometers are expensive, figuring out a cheaper alternative for these experiments is also important as many school districts may not have the funds to purchase one. Furthermore, the researchers are looking for ways to make this experiment more accessible, engaging, and accurate.
Cyanobacteria
Along the way, this work has been supported by the UTEX Culture Collection of Algae and the curator, Dr. Nobles, with many hours of input, feedback, advice, and of course culture samples. Because of their assistance, we are in the process of finishing up a story focused on the use and characterization of a genetically modified microbial consortium, where Oscillatoria lutea serve as a platform for other bacteria which have been engineered to serve as biosensors. This work is being led by Dominic B., who received a summer research fellowship, and Shireen S. On September 28th, Dominic will be presenting their research at the annual Fall Undergraduate Research Symposium. Current students are wrapping up this project as well as expanding into future avenues, looking for new biosensors to integrate into the community.
== Broad Host Range Kit == move this to No Longer Active Projects
The Broad Host Range Kit (BHR Kit) project was initiated as part of the UT Austin iGEM team in 2017 and 2018 with the help of Sean Leonard (a graduate student in the Barrick lab). This project seeks to identify DNA sequences that can be used to genetically modify a broad range of bacteria, including non-model organisms (organisms that are not commonly studied). Ultimately, we wish to assemble these sequences into a a single kit that can be shared with other researchers, regardless of their experience or background. This project was presented at the 2018 iGEM conference. In 2019, the project has continued in two veins. Part of the research is being continued in the Barrick lab by Eleanor Y. and Bibiana T., Microbe Hacker alumni from 2018. They are focused on DNA sequences called "origins of replication" and their poster on the BHR Kit won the Thermo Fisher Scientific Award for Excellence in Biological Sciences at the Undergraduate Research Forum, here at UT, in Spring 2019. The other part of the project continues within the Microbe Hackers lab. Here, we are focused on creating more "part plasmids", sequences of DNA that can be broadly used, and we are attempting integrate more DNA sequences into our kit through various avenues of research. Future directions in the stream include completing the kit, demonstrating its utility, and communicating with others who would like to have access to these DNA sequences. In particular, we are focused on building out the number of reporter genes we have, with known expression levels, and eventually we will incorporate more origins of replication into our kits as well. Additionally, this project collaborates frequently with other researchers or with our other projects, as the members of the BHR Kit often work with DNA sequences that are useful for a variety of purposes. One of our students, Ryan B., recently wrote a blog post for Benchling describing the 2018 iGEM team's project and the BHR Kit.
Evolutionary Stability
The Evolutionary Stability (EvoStab) project studies the stability of genetic devices (DNA sequences) over time. These devices normally create a "burden" on cells. Overtime, cells will mutate to reduce their burden. As this happens, cells with reduced burden grow more quickly, which is bad for us researchers. As synthetic biologist, our whole goal is to reprogram cells to perform functions or tasks that they don't normally perform. However, those functions create burden, and thus, overtime, the cells will evolve to get rid of that function. Hence, the need to study "evolutionary stability". The EvoStab project studies burden, mutation rates, and how to make genetic devices more stable (less prone to mutating and being lost in the population) over time.
This project was initiated by the 2015 iGEM team, and has continued as a stream research project since then. The UT Austin iGEM team is working on a related project, and we collaborate with them. There are currently several directions this project is moving in. One of those subprojects, focusing on stabilizing "SYFP2" will be described by Anna B. when she presents at the Fall Undergraduate Research Symposium on September 28th. The other subprojects are focusing on determining the rates of mutation for specific DNA sequences, and to what extent the mutations are present within a population over time.
Bee Project
Currently under edit. Check for accuracy. -Tarun
Bees are currently under danger due to Colony Collapse Disorder, which is when a beehive loses many of its worker bees. To help mitigate this issue, the Bee Project was started to study the function of genes in the bee genome. Bombus impatiens is the species selected for this project. Functional genomics is used to achieve this goal. Functional genomics is the study of gene functions and interactions. The project involves designing dsRNA plasmids capable of knocking down the targeted gene in B. impatiens in a process called RNA interference (RNAi). These plasmids will be stored and replicated in S. alvi , a native bee gut bacteria.
Currently, PCR products for 7 genes have been successfully produced, as well as some part plasmids and assembly plasmids. Assembly plasmids is a plasmid made by ligating multiple part plasmids together. The Bee Project members are currently troubleshooting to yield PCR products for genes without successfully PCR products. This project is being funded by the Bee Project Grant.
No Longer Active Research Projects
Over the years, we have had many projects. Some projects have seen success after many attempts. Other projects, despite our best efforts though have not gone as we originally planned. Below are a number of retired projects. Some of these projects were the inspiration for starting other areas of research or have been folded into active projects.
- Kombucha - Not Active. The main project of our 2016 iGEM team, Kombucha is a "brewed beverage", made by yeast and bacteria. We were attempting to isolate and genetically engineer these organisms in hopes of creating a "designer kombucha". After some exciting initial success in 2016, it became difficult to engineer these organisms, and as our main Kombucha experts graduated in Spring 2019, we decided to put the project on the shelves. The "'Bucha Bunch" will live on in memory.
- Mars/UV - Not Active. One of the main projects of our 2018 iGEM team, the Mars/UV project sough to genetically modify organisms for one of two purposes. First, to serve as either bacterial sunscreen, preventing harmful UV from reaching your skin. Second, to help colonize Mars by engineering the bacteria to be better suited for living on Mars, dealing with hostile growing conditions, including UV, cold temperatures, and perchlorates in the soil.
- pH sensors - Not Active. One of our earlier projects in 2015, this project sought to create reporters that would respond to differing pH conditions. Eventually, this project morphed into our Chromoproteins project, which was most active in 2017-2018. The chromoproteins project was folded into our BHR Kit project in Sp2019.
- Neonicotinoids - Not Active. One of our first 2015 projects. This ambitious project sought to isolate bacteria that could degrade neonicotinoid pesticides, in hopes of finding genes that could degrade these molecules. The goal was to then engineer bacteria to protect honeybees, by establishing genetically modified (probiotic) bacteria inside of bee guts to help them survive against pesticides.
- Broad Host Range Kit - Not Active.
Additionally, we are considering starting a new project or two in 2020, but we won't know for sure until sometime in 2020. Normally, new projects will start at the end of spring, with an emphasis on working on the projects during the summer.
Course and Community Related Information
Microbe Hackers is the "synthetic biology" research stream. However, our experiments and research can also be broadly classified as molecular biology, genetic engineering, and microbiology. In terms of concepts covered, we deal a lot with genetics, molecular biology, and microbiology. A lot of our course material overlaps with genetics and some overlaps with introductory biology. So, if you take genetics after our stream, then you can hopefully use a lot of what you learn in this FRI stream when you take genetics. And if you are currently taking genetics, then hopefully it will make your semesters with us easier.
Our course material also attempts to cover the most recent news in the field of synthetic biology and how it affects society. This includes CRISPR and the issues dealing with genetic engineering in humans, GMO foods, and how should cutting edge technology be communicated to the public in a way that inspires open dialogue and respect. In 2019, we have discussed in depth CRISPR babies and GMO mosquitoes, among other topics. Since 2019, we have discussed and read articles about COVID-19, in addition to the previous subjects listed.
The progression of the Microbe Hackers research lab is as follows: During the spring students learn concepts and techniques before being assigned to projects (with their input). Students can stay and conduct research in the summer. Students return in the fall, focused on research and the development of higher level skills: critical thinking, project management, presentations, data analysis. We also incorporate many of these skills into our spring curriculum, but not to the same degree as in the fall.
For Spring 2023
Tarun replaced instances of '2020' with '2023'
- The Lecture portion of this FRI stream should be listed as Mondays 3pm - 5pm. Only ~1 hr of this will be used for lecture. The other hour will be used as a discussion/review/technique time hour for students who want to receive extra exposure to concepts or to the lab prior to beginning that week's lab.
- The Lab portion of this FRI stream will be determined during the first week of class based on student schedules. You will have a 4 hour lab period that meets once a week during the first part of the course. Later, you will be able to schedule your own lab time.
- There will also be a weekly discussion time. Attendance is strongly suggested, but not required. You are recommended to sign up for a discussion time prior to your lab time each week!
- The stream offers CH204 credit.
NOTE: For Spring 2023 you must take the course for CH204 credit. You can take our second semester course for BIO206L or CH369K/BIO377 credit in the fall. Thus, if you stay with the stream for a full year, you can cover both your CH204 and BIO206L required courses through FRI, which means you do research for two semesters instead of the standard (boring?) intro labs.
If you have other questions, please contact Dr. Mishler. My contact information can be found on the FRI website page that brought you here.
Fall 2022 Open House
Tarun replaced instances of '2019' with '2022'
If you are interested in hearing more about the lab or our research, visit us during the Fall 2019 open house or contact Dr. Mishler.
Our Fall 2022 Open House consists of a tour of the lab that lasts approximately 30 minutes. During this tour a mentor and Dr. Mishler will introduce you to the stream, some of the research we conduct, and answer questions. Each tour starts promptly at the time indicated. Please show up 5 minutes early and wait in the hallway near the lab door.
NOTE: You must sign up for a tour using this link: Sign up link. Tours will consist of no more than 15 students at once. This link is broken, we need to replace
You will be asked to sign in before the tour starts. If your name is not on the list of attendees, you will be asked to come back later. Please sign up well in advance, if you are interested in our stream.
Course Photos
Microbe Hackers 4 Life
Microbe Hackers 4 Life is the title I give to students who join us in the spring, and then stay with the stream as mentors... until they graduate from UT Austin. In 2018, our first Microbe Hacker 4 life graduated. In 2019, five more students received this title. My appreciation for and the entire community's appreciation for these students is hard to fully explain. These rare students have spent countless hours researching and/or mentoring younger students. They have taught our younger students techniques, gone over concepts, given feedback on papers, helped to run the lab, helped to clean the lab, and most importantly established a friendly, nurturing environment for future Microbe Hackers. Upon graduating, these students receive a small token of my appreciation for their efforts, normally an item somehow related to their time with us.
All of our mentors do a tremendous service for our community, whether they are with us for one semester or five. On behalf of the entire Microbe Hackers lab, thank you all so much for being with us!
Summer 2019 Researchers and Trivia
Every year, the Microbe Hackers lab is open during the summer. We normally start immediately after Memorial day and go for 8 weeks, ending in mid-to-late July. It is Professor Mishler's stated goal to find as much space as possible for as many students as possible to conduct research with us over the summer. A few students received summer fellowships, to help them stay, but most students volunteer during this time.
In 2019, we had 16 of our freshmen stay with us during the summer, in addition to 8 of our returning mentors who conducted research and helped the younger students around the lab. Students are in lab for at least 20 hrs per week during this 8 week period. We have weekly meetings, focused on developing presentation skills, as well as other important skills: such as making professional figures and analyzing data.
Every Friday, the students organize a lunch, either on the patio outside of lab or at a nearby restaurant. In addition, the lab had our first ever "trivia" event at the end of the summer. Teams were formed based on the benches students worked at and answered questions relating the our lab, science, Austin, and popular culture.
LinkedIn Page and Alumni
To our alumni, please contact us! I love it when you drop by or send me letters or e-mails. Hearing about what you are up to or the challenges you face inspires me. Seriously, they do. Being with us was only one part of your time at UT Austin, and only a small part of your overall life, but if we can make a positive impact on your education, career, or life goals, we absolutely want to.
Recently, we started a Microbe Hackers LinkedIn page! Please join, follow, or do whatever you are supposed to do with LinkedIn... I really don't understand it, but the mentors do a great job updating and maintaining the site. Thank you Mah-ro (creator) and Michael and Roshni (current maintainers)!
Microbe Hackers Publications and Presentations
Publications [update in progress]
Need to import actual references.
- Caffeinated Coli 2.0 [in press]
- Synthetic genome defenses against selfish DNA elements stabilize engineered bacteria against evolutionary failure.
- Bacterial Production of Gellan Gum as a Do-It-Yourself Alternative to Agar.
- Rapid and Inexpensive Evaluation of Nonstandard Amino Acid Incorporation in Escherichia coli
- Reproducibility of fluorescent expression from engineered biological constructs in E. coli.
- Predicting the genetic stability of engineered DNA sequences with the EFM calculator.
Poster Presentations [update in progress]
So many!!! Need to decide how to incorporate this information.
iGEM Competition
Tarun replaced '2019' with '2022. No further updates made.
iGEM 2022 Team : NEEDS TO BE UPDATED. NOT CURRENT
The 2022 iGEM team application period is NOT currently open.
Our iGEM team is composed solely of UT Austin undergraduates. These students develop a new project each year, and work on it from Spring to Fall under the guidance of UT professors and graduate students. At the end of this timeline, representatives go to Boston and present their research at an international meeting. Applications are due in "fall", and the new members are selected by the end of the semester.
The Microbe Hackers FRI stream also feeds directly into the UT Austin iGEM team, which is led by Professor Mishler and Professor Barrick. Students who conduct research with us during the year, and especially in the summer, are able to participate in the annual project. Every fall, a handful of students are selected to represent UT Austin at this international academic event that features hundreds of teams from around the world, 90% of which are comprised solely of undergraduates conducting research in synthetic biology. Below are photos of previous teams in Boston.
NOTE: The next application period will open in the near future. If you are interested, please feel free to inquire for details.
Previous iGEM teams
Here are our team pages from 2021, 2020, 2019, 2018, 2017, 2016, 2015, and 2014.