Difference between revisions of "CH391L/S14/SmallRNAs"
(→sRNAs in Synthetic Biology) |
|||
| Line 18: | Line 18: | ||
[[File:Figure2review.png|thumb|right|200 px|Figure 2: Composability of sRNAs as a strategy for the synthesis of artificial RNA devices. sRNAs are regulators of high modularity. An sRNA-based regulator can be broken down in two main parts: a sensor (target binding domain) and a stabilizer (that can include an Hfq-binding site and the transcriptional termination domain). In the context of a genetic device, the sRNA binds an mRNA target. In this case, the 5′ UTR of the target mRNA acts as an adaptor that transmits the signal to the gene reporter actuator. The combination of the sRNA and mRNA target comprises a functional synthetic device.<cite>Vazquez2013</cite>]] | [[File:Figure2review.png|thumb|right|200 px|Figure 2: Composability of sRNAs as a strategy for the synthesis of artificial RNA devices. sRNAs are regulators of high modularity. An sRNA-based regulator can be broken down in two main parts: a sensor (target binding domain) and a stabilizer (that can include an Hfq-binding site and the transcriptional termination domain). In the context of a genetic device, the sRNA binds an mRNA target. In this case, the 5′ UTR of the target mRNA acts as an adaptor that transmits the signal to the gene reporter actuator. The combination of the sRNA and mRNA target comprises a functional synthetic device.<cite>Vazquez2013</cite>]] | ||
| − | sRNAs are highly composable, (composability is the ability of a system to berak down in units due to the system modularity and recombine in different configurations to satisfy specific human requirements), tunable and their | + | sRNAs are highly composable, (composability is the ability of a system to berak down in units due to the system modularity and recombine in different configurations to satisfy specific human requirements), tunable and their orthogonality can be designed a priori. In general, a variety of strategies have been used to synthetize sRNAs that include rational design, model-driven computational design, in vivo and in vitro molecular evolution and selection and, harvesting of natural parts <cite>Vazquez2013</cite>. Efforts have focused on preserving the sRNA scaffold, which includes an Hfq domain and a transcriptional terminator, and engineering the binding domain (see Figure 2 for a schematics of sRNA breakdown). |
=== Designing a synthetic sRNA === | === Designing a synthetic sRNA === | ||
| − | [[File:Figure3review.png|thumb|left|200 px|Figure 2: Artificial sRNA screening strategy and library design. (a) Schematic illustration of the artificial sRNA screening strategy. A reporter vector with the target mRNA leader sequence fused to gfpuv is cotransformed with a partially randomized artificial sRNA expression library and plated on agar plates. | + | [[File:Figure3review.png|thumb|left|200 px|Figure 2: Artificial sRNA screening strategy and library design. (a) Schematic illustration of the artificial sRNA screening strategy. A reporter vector with the target mRNA leader sequence fused to gfpuv is cotransformed with a partially randomized artificial sRNA expression library and plated on agar plates. Colonies with weaker fluorescence are picked and characterized. (b) Artificial sRNA library based on the Spot42 sRNA scaffold (yellow box). The antisense domain in Spot42 (identified for galK) is shown in gray, and the bases that were shown to interact with Hfq are indicated in bold.5 Degenerate bases (N) were inserted between the vector-derived sequence (50-ACUCGAG-30) and the sRNA scaffold.<cite>Sharma2012</cite>]] |
| − | Colonies with weaker fluorescence are picked and characterized. (b) Artificial sRNA library based on the Spot42 sRNA scaffold (yellow box). The antisense domain in Spot42 (identified for galK) is shown in gray, and the bases that were shown to interact with Hfq are indicated in bold.5 Degenerate bases (N) were inserted between the vector-derived sequence (50-ACUCGAG-30) and the sRNA scaffold.<cite>Sharma2012</cite>]] | + | |
| − | Three factors likely influence sRNAs ability to regulate gene expression: kinetics of binding, extension and energy of binding as well as the types and number of mRNAs that a given sRNA can bind. Based on these factors Sharma et al.<cite>Sharma2012</cite> developed a high-throughput strategy for the engineering of synthetic sRNAs. In their approach, the Hfq domain was left unchanged and a library of randomized binding domains was generated. A natural 5’ UTR was fused to a reporter gene (GFP) and the researchers selected for the repression of this gene. They were able | + | Three factors likely influence sRNAs ability to regulate gene expression: kinetics of binding, extension and energy of binding as well as the types and number of mRNAs that a given sRNA can bind. Based on these factors Sharma et al.<cite>Sharma2012</cite> developed a high-throughput strategy for the engineering of synthetic sRNAs. In their approach, the Hfq domain was left unchanged and a library of randomized binding domains was generated. A natural 5’ UTR was fused to a reporter gene (GFP) and the researchers selected for the repression of this gene. They were able to successfully identify sRNA candidates that repress ompF and fliC mRNAs. Interestingly, the authors observed that the artificial constructs repressing the ompF exhibit important similarities in the features shown by the natural ompF repressor, the sRNA MicF (Figure 3). A recent work studied the free-energy of the complex sRNA-mRNA and found an important correlation between structure-function in sRNAs. Hao et al. <cite>Hao2011</cite> generated numerous mutants of the sRNA RyhB and tested in vivo their gene control function. They concluded that when using a thermodynamic model to compute the free-energy of the mRNA-sRNA complex, these values exponentially correlated to the gene silencing strengths shown by the mutants. |
=== sRNAs in metabolic engineering === | === sRNAs in metabolic engineering === | ||
| − | As aforementioned, sRNAs are ideal candidates for developing and alternative methodology for the combinatorial knockdown of genes in metabolic engineering. Towards these purposes, Na et al.<cite>Na2013</cite> generated a library of artificial sRNAs that target a diversity of chromosomal gene targets. Then, by a combinatorial approach they isolated a strain that was able to substantially increase cadaverine production and tyrosine production. This approach is generalizable to other bacterial strains. The strategies proposed by the authors possess important advantages over traditional gene knockouts methodologies due to the ability to fine-tune gene silencing, target multiple genes, easy-implementation and the ability to modulate gene expression without modifying those genes. These strategies avoid the burdensome generation of strain libraries. | + | As aforementioned, sRNAs are ideal candidates for developing and alternative methodology for the combinatorial knockdown of genes in metabolic engineering. Towards these purposes, Na et al.<cite>Na2013</cite> generated a library of artificial sRNAs that target a diversity of chromosomal gene targets. Then, by a combinatorial approach they isolated a strain that was able to substantially increase cadaverine production and tyrosine production. Specifically, the authors of this work selected the MicC sRNA scaffold, that includes the Hfq-binding site, and modify the binding domain by the introduction of anti-sequences of genes involved in the metabolic pathway of either cadaverine or tyrosine. Subsequently, they created a library of anti-sense RNAs and isolated the strains with higher production of the target molecules. Finally, used what they called forward engineering, to fine-tune the production optimization of these two metabolites by binding energy. They identified genes not expected to affect the titer of these metabolites but that are involved in the metabolic pathway regulation. This last realization represents a advantage over other traditional metabolic engineering approaches. In addition, this sRNA-based approach is generalizable to other bacterial strains. The strategies proposed by the authors possess important advantages over traditional gene knockouts methodologies due to the ability to fine-tune gene silencing, target multiple genes, easy-implementation and the ability to modulate gene expression without modifying those genes. These strategies avoid the burdensome generation of strain libraries. |
| − | As it can be confirmed from table 1, there are very few examples of the use of sRNAs for metabolic engineering applications. | + | As it can be confirmed from table 1, there are very few examples of the use of sRNAs for metabolic engineering applications. However, it is expected that this field will soon explode to produce numerous works and even applications aiming for more efficient strain optimization techniques for the production of biotechnologically relevant molecules. |
[[File:Table1Reviewa.png|thumb|center|1000 px|Table 1. Recent synthetic sRNAs and their (potential) applications (basic devices)<cite>Vazquez2013</cite>]] | [[File:Table1Reviewa.png|thumb|center|1000 px|Table 1. Recent synthetic sRNAs and their (potential) applications (basic devices)<cite>Vazquez2013</cite>]] | ||
Revision as of 17:26, 14 April 2014
Contents |
Bacterial small RNAs: as a potential powerful tool for metabolic engineering
Introduction
Bacterial small RNAs (sRNAs) are gene regulatory entities that range from 21 to 400 nucleotides in size. These RNAs are in charge of controlling expression of stress-response genes and thus are essential for an organism's survival under different extreme environmental conditions (e.g. nutrient availability, osmolarity, pH and temperature)[1]. The presence of these regulatory molecules appears to be ubiquitous as they have been discovered in a wide range of bacterial species [2][3]. Their high modularity and orthogonality have raised interest among synthetic biologists towards the construction of sRNA-like devices. In addition, sRNA capacity to simultaneously target single or multiple genes with high specificity has enabled the vision of sRNAs as a powerful tool for metabolic engineering applications.
Bacterial small RNAs
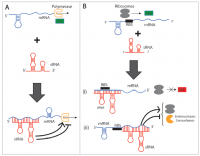
sRNAs can be classified as cis-encoded and trans-encoded. The former refers to those that are transcribed from the complementary strand of the genes that they target. This class represents the minority of the sRNAs that have been identified up to now. Additionally, cis-encoded sRNAs usually exert a tight control over a single target messenger RNA (mRNA). In contrast, trans-encoded sRNAs are transcribed from loci in the genome that are distant from where their mRNA targets are encoded. This class accounts for the great majority of sRNAs discovered to date. An astonishing feature is that these molecules can bind their mRNA partners by a minimal base-pairing requirement (8-9 nucleotides)[1]. Lastly but more importantly, this class of sRNAs can interact with multiple mRNAs[5]. This property, in turn, enables the potential application of combinatorial gene knockdown in metabolic engineering.
Trans-encoded sRNAs can target proteins in addition to mRNAs; an example of that are sRNAs such as CsrB/C and 6S RNA. When controlling mRNA expression this class of sRNAs uses a diversity of mechanisms. They can (1) base-pair to their target mRNAs to enhance or attenuate transcription (Figure 1A), (2) directly block (Figure 1B i), or indirectly enhance or inhibit translation (Figure 1B ii), (3) sequester proteins (not shown), or (4) directly lead to mRNA and protein degradation (Figure 1B iii). This article will exclusively focus on those sRNAs that are trans-encoded and only target mRNAs. Hereafter, they will be referred simply as sRNAs. This class of sRNAs, as aforementioned, accounts for the majority of discovered sRNAs and can target multiple genes. Consequently, these sRNAs have attracted much interest among the Synthetic Biology community as it will be shown in the remainder of this article.
A particular feature that this class of sRNAs exhibits is the interaction with a major chaperone protein called Hfq. These interactions have been mainly observed in gram-negative bacteria. Hfq action leads to the stability of sRNAs, assists their binding to target mRNAs and stabilizes interactions sRNA-mRNA[1]. Recent reports propose that Hfq can also exert negative regulation by delivering the sRNA-mRNA complex to the degradosome [3]. By engineering Hfq interaction, gene expression control could potentially be greatly improved since the gene repression dynamic range is enhanced. In addition, the introduction of Hfq domains into an already constructed sRNA-like device could bring about a very valuable increase in its gene silencing capabilities[6].
sRNAs in Synthetic Biology
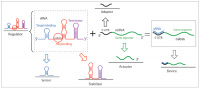
sRNAs are highly composable, (composability is the ability of a system to berak down in units due to the system modularity and recombine in different configurations to satisfy specific human requirements), tunable and their orthogonality can be designed a priori. In general, a variety of strategies have been used to synthetize sRNAs that include rational design, model-driven computational design, in vivo and in vitro molecular evolution and selection and, harvesting of natural parts [4]. Efforts have focused on preserving the sRNA scaffold, which includes an Hfq domain and a transcriptional terminator, and engineering the binding domain (see Figure 2 for a schematics of sRNA breakdown).
Designing a synthetic sRNA
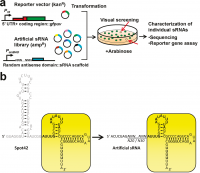
Three factors likely influence sRNAs ability to regulate gene expression: kinetics of binding, extension and energy of binding as well as the types and number of mRNAs that a given sRNA can bind. Based on these factors Sharma et al.[7] developed a high-throughput strategy for the engineering of synthetic sRNAs. In their approach, the Hfq domain was left unchanged and a library of randomized binding domains was generated. A natural 5’ UTR was fused to a reporter gene (GFP) and the researchers selected for the repression of this gene. They were able to successfully identify sRNA candidates that repress ompF and fliC mRNAs. Interestingly, the authors observed that the artificial constructs repressing the ompF exhibit important similarities in the features shown by the natural ompF repressor, the sRNA MicF (Figure 3). A recent work studied the free-energy of the complex sRNA-mRNA and found an important correlation between structure-function in sRNAs. Hao et al. [8] generated numerous mutants of the sRNA RyhB and tested in vivo their gene control function. They concluded that when using a thermodynamic model to compute the free-energy of the mRNA-sRNA complex, these values exponentially correlated to the gene silencing strengths shown by the mutants.
sRNAs in metabolic engineering
As aforementioned, sRNAs are ideal candidates for developing and alternative methodology for the combinatorial knockdown of genes in metabolic engineering. Towards these purposes, Na et al.[9] generated a library of artificial sRNAs that target a diversity of chromosomal gene targets. Then, by a combinatorial approach they isolated a strain that was able to substantially increase cadaverine production and tyrosine production. Specifically, the authors of this work selected the MicC sRNA scaffold, that includes the Hfq-binding site, and modify the binding domain by the introduction of anti-sequences of genes involved in the metabolic pathway of either cadaverine or tyrosine. Subsequently, they created a library of anti-sense RNAs and isolated the strains with higher production of the target molecules. Finally, used what they called forward engineering, to fine-tune the production optimization of these two metabolites by binding energy. They identified genes not expected to affect the titer of these metabolites but that are involved in the metabolic pathway regulation. This last realization represents a advantage over other traditional metabolic engineering approaches. In addition, this sRNA-based approach is generalizable to other bacterial strains. The strategies proposed by the authors possess important advantages over traditional gene knockouts methodologies due to the ability to fine-tune gene silencing, target multiple genes, easy-implementation and the ability to modulate gene expression without modifying those genes. These strategies avoid the burdensome generation of strain libraries.
As it can be confirmed from table 1, there are very few examples of the use of sRNAs for metabolic engineering applications. However, it is expected that this field will soon explode to produce numerous works and even applications aiming for more efficient strain optimization techniques for the production of biotechnologically relevant molecules.

A robust gene expression control device inspired on sRNAs

Isaacs et al.[10] developed a riboregulator system showing an enhanced dynamic range. This riboregulator design is inspired on the DsrA-RpoS sRNA system (Figure 4). This system has pioneered the field of rational design of sRNA-like systems and seeded a variety of applications based upon this same device. More recently, this cr-taRNA system has been used to test the influence of the Hfq assistance. Sakai et al.[6] introduced a Hfq domain into the taRNA and found improved results in gene expression control suggesting that in vivo Hfq enhances the inherent sRNA regulatory capacity.
sRNA-like iGEM projects
The Denmark Technical University team in 2011 used a bioinformatics approach to confirm the structural features present in an sRNA e.g. binding domain, Hfq domain, transcription terminator and linker region. They investigated the sRNA system chitobiose that requires the presence of another sRNA called trap-RNA (in this case chiXR) to release the silencing imparted by chiX on its target mRNA chiP. This work represents an interesting confirmation experiment of what had been already reported in the literature since they inserted chiP in a plasmid a showed that its expression was regulated by chiX and when changing the complementary binding region the regulation is removed.
Other teams such as the Ocean University of Chine iGEM 2012 team aimed to develop a decision-making device based on sRNA regulation to predict when red tide is going to happen. In another example, Uppsala University iGEM 2012 team constructed synthetic sRNAs that can down regulated antibiotic resistance genes by engineering the binding domain of the sRNA Spot42.
References
Error fetching PMID 20980440:
Error fetching PMID 21925377:
Error fetching PMID 23362267:
Error fetching PMID 23651005:
Error fetching PMID 21742981:
Error fetching PMID 23334451:
Error fetching PMID 24356572:
Error fetching PMID 15208640:
Error fetching PMID 24328142:
- Error fetching PMID 15487940:
Comprehensive review on bacterial small RNAs - Error fetching PMID 20980440:
A more recent review on bacterial small RNAs. - Error fetching PMID 21925377:
Another recent review on bacterial small RNAs. - Error fetching PMID 24356572:
A thorough review on synthetic regulatory RNAs. - Error fetching PMID 23362267:
A review on sRNA negative regulation. - Error fetching PMID 24328142:
Effect of Hfq domain introduction into a synthetic sRNA. - Error fetching PMID 23651005:
High-throughput method for the engineering of sRNAs. - Error fetching PMID 21742981:
sRNA structure-function relationship. - Error fetching PMID 23334451:
sRNAs in metabolic engineering. - Error fetching PMID 15208640:
A robust sRNA-inspired riboregulator. - [http://2011.igem.org/Team:DTU-Denmark/Project
sRNA system with a trap-RNA for chitibiose control.