Gene Therapy versus Human Genome Editing (Biotechnology and Society 2016)
From SynBioCyc
Revision as of 03:58, 31 October 2016 by Jeffrey Barrick (Talk | contribs)
Glybera (Alipogene tiparvovec) gene therapy
Download Pre-Discussion Questions (MS Word)
Download Policy Czar Template (MS Word)
Everyone: Overview of gene therapy, germline modification, and genome synthesis
Required Reading/Viewing
- Gene Therapy emerges from disgrace to be the next big thing, again. Carl Zimmer Wired Science Blogs August 13, 2013.
- Engineering the Perfect Baby Antonio Regalado MIT Technology Review March 5, 2015.
Use of CRISPR for human germline editing. - Scientists reveal proposal to build human genome from scratch Kelly Servick Science June 2, 2016.
Short article describing the Human Genome Project–Write meeting.
Additional Resources
- Gene Therapy: Molecular Bandage? Educational website at the University of Utah.
Especially, check out the "Tools of the Trade" and "Space Doctor" links. - Read Chapter 5: Ethical and Social Problems of Gene Therapy
Access from on-campus to read.
CRISPR Germline Modification Czar
Required Reading/Viewing
- Everything You Need to Know About CRISPR, the New Tool that Edits DNA Sarah Zhang Gizmodo May 6, 2015.
- Chinese scientists genetically modify human embryos Cyranoski and Reardon. Nature News April 22, 2015.
- Editing Human Embryos: So This Happened Carl Zimmer National Geographic Blogs: The Loom April 22, 2015.
- Lanphier, E., Urnov, F., Haecker, S.E., Werner, M., and Smolenski, J. (2015). Don’t edit the human germ line. Nature 519: 410–411.
- Baltimore, B.D., Berg, P., Botchan, M., Carroll, D., Charo, R.A., Church, G., Corn, J.E., Daley, G.Q., Doudna, J.A., Fenner, M., et al. (2015). A prudent path forward for genomic engineering and germline gene modification. Science 348:36–38.
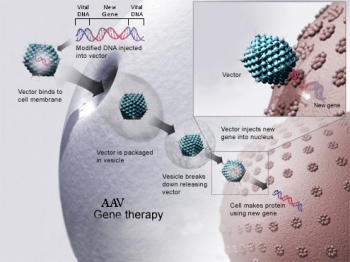
Gene Therapy Czar
Required Reading/Viewing
- Hacein-Bey-Abina, S., Garrigue, A., Wang, G.P., Soulier, J., Lim, A., Morillon, E., Clappier, E., Caccavelli, L., Delabesse, E., Beldjord, K., et al. (2008). Insertional oncogenesis in 4 patients after retrovirus-mediated gene therapy of SCID-X1. J. Clin. Invest. 118:3132–3142.
Scan for the major take-home message. - Bryant, L.M., Christopher, D.M., Giles, A.R., Hinderer, C., Rodriguez, J.L., Smith, J.B., Traxler, E.A., Tycko, J., Wojno, A.P., and Wilson, J.M. (2013). Lessons learned from the clinical development and market authorization of Glybera. Hum. Gene Ther. Clin. Dev. 24:55–64.
Chronicles the development of the first commercially available gene therapy treatment for Familial Lipoprotein Lipase Deficiency (LPLD). - Glybera - commercial gene therapy product.